
Breaking down the microbiology world one bite at a time
Wired for Clean Energy: How unique bacteria make electricity-conducting wires
This post is written by guest author Audrey Burnim
We often take for granted how a quick flip of a switch can illuminate a room, but there is a complicated, highly engineered process for the movement of electrons from power source to lightbulb. Recently, researchers at Yale University investigated how a common soil bacterium called Geobacter can produce electricity using proteins and metal ions. This unique ability allows them to thrive in environments with limited oxygen, such as contaminated soils and sediments. One of the key components of this electron transfer process is the formation of nanowires, which serve as conductive bridges for the transfer of electrons from the bacteria to external electron acceptors.
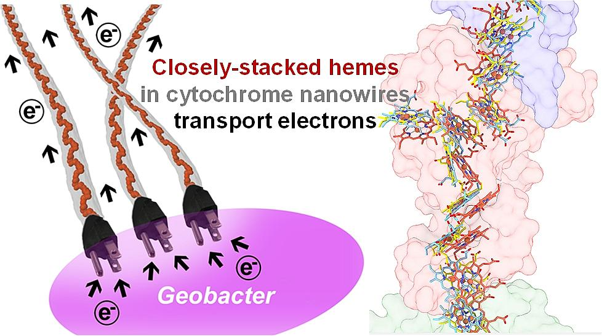
Researchers have been interested in this family of bacteria due to their unique ability to survive without oxygen and eat methane. Fascinatingly, the metabolism of the bacteria also moves electrons outside of their cells, producing electrical power in a process known as extracellular electron transport. Methane is a key contributor to greenhouse gasses that are rapidly accelerating climate change, so it is possible for these bacteria to be a potential solution for generating energy and ameliorating the climate crisis.1 How these proteins organize their own machinery in a way that allows the formation of a molecular wire, shuttling electrons efficiently—just like wires that we use—has been largely a mystery until this work by Yangqi Gu and coworkers. One long-term goal of this molecular-level exploration is so scientists can understand the metabolism of these microbes at a deeper level to engineer bacteria that are more efficient at eating methane.
In this recent article in Nature Microbiology called “Structure of Geobacter cytochrome OmcZ identifies mechanism of nanowire assembly and conductivity,” the researchers used cryogenic electron microscopy (Cryo-EM) and other experiments to look at how Geobacter sulfurreducens CL-1 makes these wires at the molecular level and how they generate electrical current. They found that these nanowires were made by precisely arranging metal-containing molecules, called heme compounds (or haem compounds). Specifically, these molecules contain iron (Fe) at their center. Our bodies move iron and oxygen around, using the same heme compounds, and they make our blood red!2 These heme molecules are held precisely in place by proteins called “OmcZ”. OmcZ is classified as a cytochrome, literally meaning “cellular pigment”, because the structures have a characteristic set of observable colors based on how the molecule absorbs light.
The researchers discovered that OmcZ is the only protein that is essential for the formation of electrical current in Geobacter sulfurreducens biofilms. Biofilms refer to how Geobacter sulfurreducens interact with each other.3,4 They form large aggregates and matrices of their cells and extracellular components, in this case forming a large complex of nanowires. The density of G. sulfurreducens in the biofilm was very high, which contributed to the high amount of electricity observed.
Cryogenic electron microscopy (cryo-EM) was used to visualize these nanowires. Cryo-EM is a powerful—and Nobel Prize-winning5—imaging technique that allows researchers to take samples of proteins, fire electrons at them, and reconstruct what their electron density looks like, allowing a 3D molecular model to be built. A simplified representation of an example workflow is shown below.
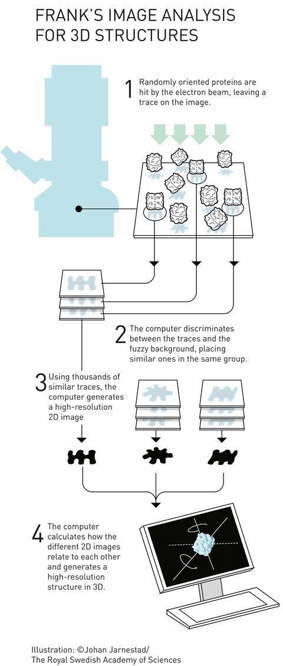
This 3D model that resulted from this work (shown on the left of the figure at the beginning of this article) gave a detailed picture of how individual pieces of this nanowire are built, and the researchers can then build a story from this picture.
Furthermore, the researchers found that the organization of the nanowire network can re-arrange in different biochemical environments, indicating the adaptability of OmcZ nanowires to different conditions. This versatility may explain why Geobacter species are able to thrive in diverse environments with varying electron acceptors.
In addition to the structural findings, the researchers also uncovered the role of a serine protease, a type of enzyme, in controlling the assembly of OmcZ monomers into nanowires. This protease was found to be crucial in regulating the formation and stability of OmcZ nanowires. This discovery provides insights into the intricate mechanisms underlying the assembly of OmcZ nanowires and sheds light on the complex interplay between proteins in the formation of these conductive structures.
Fascinatingly, the researchers found that both OmcZ and the serine protease are widespread in environmentally important bacteria and archaea, indicating that nanowire biogenesis may be a prevalent phenomenon across diverse species and environments. This highlights the significance of OmcZ nanowires in the microbial world and their potential implications for environmental processes, such as bioremediation and energy production.
The findings of this study have opened up new possibilities for understanding the fundamental mechanisms of electron transfer in bacteria and the potential applications of OmcZ nanowires in various fields. The high electron conductivity and adaptability of OmcZ nanowires make them a promising candidate for bioelectronic devices, such as biofuel cells and biosensors. The widespread presence of OmcZ and the serine protease in diverse bacterial species suggests that nanowire biogenesis may be a common strategy employed by bacteria for extracellular electron transport.
Link to the original post: Gu, Y., Guberman-Pfeffer, M.J., Srikanth, V. et al. Structure of Geobacter cytochrome OmcZ identifies mechanism of nanowire assembly and conductivity. Nat Microbiol 8, 284–298 (2023). https://doi.org/10.1038/s41564-022-01315-5
Featured image: Protein Nanowire Lab, Yale University.
(1) What is the greenhouse effect?. Climate Change: Vital Signs of the Planet. https://climate.nasa.gov/faq/19/what-is-the-greenhouse-effect/ (accessed 2023-04-28).
(2) Lichtin, A. Does Blood Turn Blue?. Cleveland Clinic. https://health.clevelandclinic.org/what-color-is-blood/ (accessed 2023-04-28).
(3) Biofilm. https://doi.org/10.1016/B978-0-323-85146-6.00020-6.
(4) Donlan, R. M. Biofilms: Microbial Life on Surfaces. Emerg. Infect. Dis. 2002, 8 (9), 881–890. https://doi.org/10.3201/eid0809.020063.(5) The Nobel Prize in Chemistry 2017. Nobelprize.org. https://www.nobelprize.org/prizes/chemistry/2017/press-release/ (accessed 2023-04-28).


































































































