
Breaking down the microbiology world one bite at a time
PCOS and gut microbiota on the body’s social media map
Our body is an amalgamation of a myriad of processes involving several key players. These players include internal organs, tissues, cells, hormones, and of course our body’s permanent residents: microbes. However, there is a complex social network where each entity is connected to another across several levels, much like the ones of popular social media platforms (eg: LinkedIn). One such prominent link is between the reproductive system and the intestinal microbiota.
What is PCOS?
The ovary is an organ of the female reproductive system and is instrumental in the production of egg cells in form of fluid-filled follicles. Usually, only one egg cell develops and is released for fertilization at a time. However, sometimes these developing egg cells do not mature fully and arrest in immature forms. These undeveloped cells accumulate around the edges of the ovaries. This condition is called polycystic ovarian syndrome (PCOS). There have been several studies on this topic. Since the last few decades, the connection between PCOS and gut microbiota has been studied in several laboratories worldwide.
PCOS presents itself in many shades. Worldwide, around 10% of women suffer from PCOS. 80% of these women have an additional diagnosis of insulin resistance. Insulin is a hormone that helps in controlling blood sugar levels. Insulin resistance refers to a condition when the human body cells fail to respond to insulin, thus increasing blood sugar. Women with PCOS and insulin resistance have a BMI usually above 25 and they fall in the category of overweight or obese. Such a combined diagnosis consequently leads to central obesity, impaired blood glucose levels, and in extreme cases Type-II diabetes.
Many international organizations have proposed definitions of PCOS, but the Rotterdam criteria are the most accepted to date. In 2003, a consensus workshop organized in Rotterdam suggested the definition that a person can be diagnosed with PCOS, if any 2 out of 3 criteria are met, in the absence of other factors that might cause these symptoms:
- Oligoovulation and/or anovulation
This refers to irregular ovulation or a lack of ovulation during a woman’s menstrual cycle.
- Excess androgen activity
Androgens are a set of steroid hormones responsible for maintaining male characteristics in vertebrates. Excessive facial hair falls under this category.
- Polycystic ovaries
12 or more underdeveloped follicles arranged at the periphery of the ovaries should be seen on an ultrasound examination.
In 2006, these criteria were further tightened around androgen excess. The change in the criteria can be attributed to the fact that the symptoms are largely inconsistent in women suffering from PCOS.
PCOS in animal models
In mice, PCOS can be induced by using natural hormones like dihydrotestosterone (DHT). DHT can be converted to testosterone by the action of certain enzymes. Testosterone is a primary androgen in mammals.
Do PCOS symptoms affect the gut microbiota?
In an earlier study by a research team from Sydney, Australia, it was shown that macronutrients like protein, carbohydrate, and fat in diet do not significantly alter the metabolic traits (e.g.: insulin resistance), but restore the ovulation in a PCOS-like mouse model. In a recent study, the same team explored whether the diet intervention alters the gut microbiota in a PCOS-like mouse model. They administered DHT to mice for at least 2 weeks to induce PCOS-like symptoms which include hormonal changes, reproductive changes like anovulation, and metabolic changes like increased insulin resistance. Both control mice and PCOS-like mice were given 10 different types of diets with varying concentrations of macronutrients and fecal pellets were collected (Image 1A).
The results indicate that diet itself does not impact PCOS in terms of overall gut microbial composition. However, on clustering these samples using sophisticated mathematical methods, scientists found that there is a significant difference between the two groups. The dominant taxa that were present in different abundance (relative amounts) in the two groups belonged to Bacteroides genus, especially Bacteroides acidifaciens. The latter prevents the onset of obesity as proven in previous studies based on mouse models. The abundance of Bacteroides acidifaciens was the highest at 5% protein, 20% fat, and 75% carbohydrate in both groups.
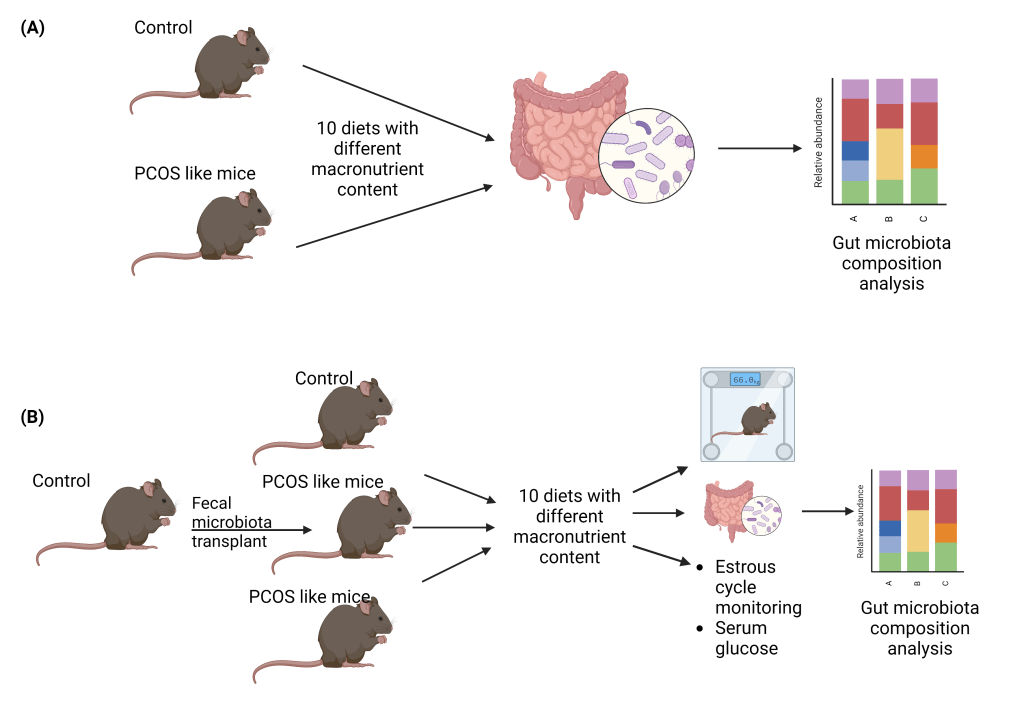
Image 1: Experimental setup for two experiments. (A) Control healthy mice and PCOS-like mice were given 10 different diets, followed by intestinal content sampling and gut microbiota analysis. (B) Fecal microbiota from control healthy mice was transplanted to PCOS-like mice. Control healthy mice, PCOS-like mice, and fecal microbiota transplanted PCOS-like mice were given 10 different diets, followed by intestinal content sampling and gut microbiota analysis. Image created by the author in biorender.com.
What is the impact of fecal microbiota transplant on PCOS?
The results from gut microbiota analysis of control and PCOS-like mice sparked many more questions, one of which was whether transplanting the gut microbiota of healthy control mice in the PCOS-like mice would impact their gut microbial dynamics and the symptoms of PCOS. With this question in mind, the researchers conducted another experiment where they transplanted the diluted fecal material (fecal microbiota transplant (FMT)) of control mice into PCOS-like mice at regular intervals for 2 weeks (total 6 transplants). After 2 weeks, there was no difference in the body weight of either control healthy, control PCOS-like mice, or FMT-treated PCOS-like mice (Image 1B). Researchers did not observe any difference between the estrous cycle (similar to the menstrual cycle in humans) of mice in the three groups. Neither were there any differences observed for the serum glucose levels during fasting and insulin levels after feeding.
While the metabolic traits of PCOS were not affected by the fecal microbiota transplant, there was a change in the gut microbiota. A decrease in the number of different bacterial taxa was observed in FMT-treated PCOS-like mice compared to control PCOS-like mice. In addition, by sophisticated clustering methods, researchers found that the FMT-treated PCOS-like mice clustered in the vicinity of control healthy mice as compared to control PCOS-like mice. This means that the abundances of individual bacteria have an impact on PCOS symptoms.
Image 2: Connection between gut microbiota and polycystic ovarian syndrome. Image created by the author on biorender.com (Featured image and in-text image)
What does this mean for women affected by PCOS?
Overall this research highlights the role of the specific composition of macronutrients in shaping the gut microbiota of women suffering from PCOS. While the diet itself is not enough for reducing the metabolic and endocrine characteristics of PCOS, it still affects the reproductive features and restores ovulation. However, therapies like fecal microbiota transplant are still far from their effective application for ameliorating PCOS.
There are still several years left before science fully decodes the connection between PCOS and the gut microbiota, yet these links are unravelling gradually with each new study year by year.
Featured image: Image created by the author on biorender.com


































































































