Breaking down the microbiology world one bite at a time
World microbiome day!
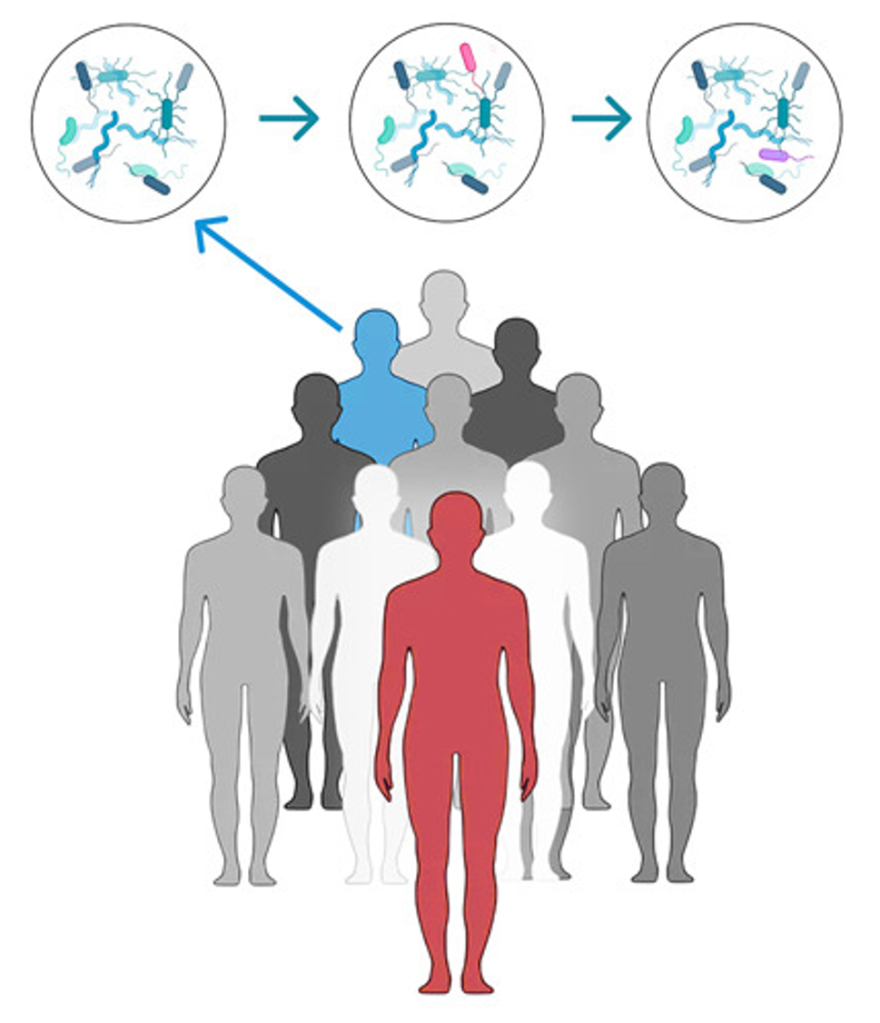
Microbiomes are not just human microbiomes, a microbiomerefers to microorganisms in a defined environment along with the environment itself (ISAPP).
Learn about what is a microbiome by the Microbiology Society.
Discover microbiome stories on MicroBites
World Microbiome Day Extravaganza: Top Science Communicators Reveal Their Microbe Moment! with Microbigals
MicroBites gave a talk on microbiomes on the Biocord network platform, check out their platform here to connect with biology enthusiasts from all over the world.
Our talk will be found in the lecture library
We’ve received many questions from you! Thank you so much for your interest in World Microbiome Day.
Some selected questions with answers can be found below.
Will there be a cure for herpes simplex virus?
Herpes simplex virus (HSV) types 1 and 2 are widespread. They are important human pathogens causing oral and genital ulcers and once you are infected, this virus will stay dormant inside your body. When it is reactivated from the dormant state, it can cause lesions again and mucosal surfaces. So far, antiviral therapy has been focused on reducing the severity of acute infections and diminishing the reactivation frequency.
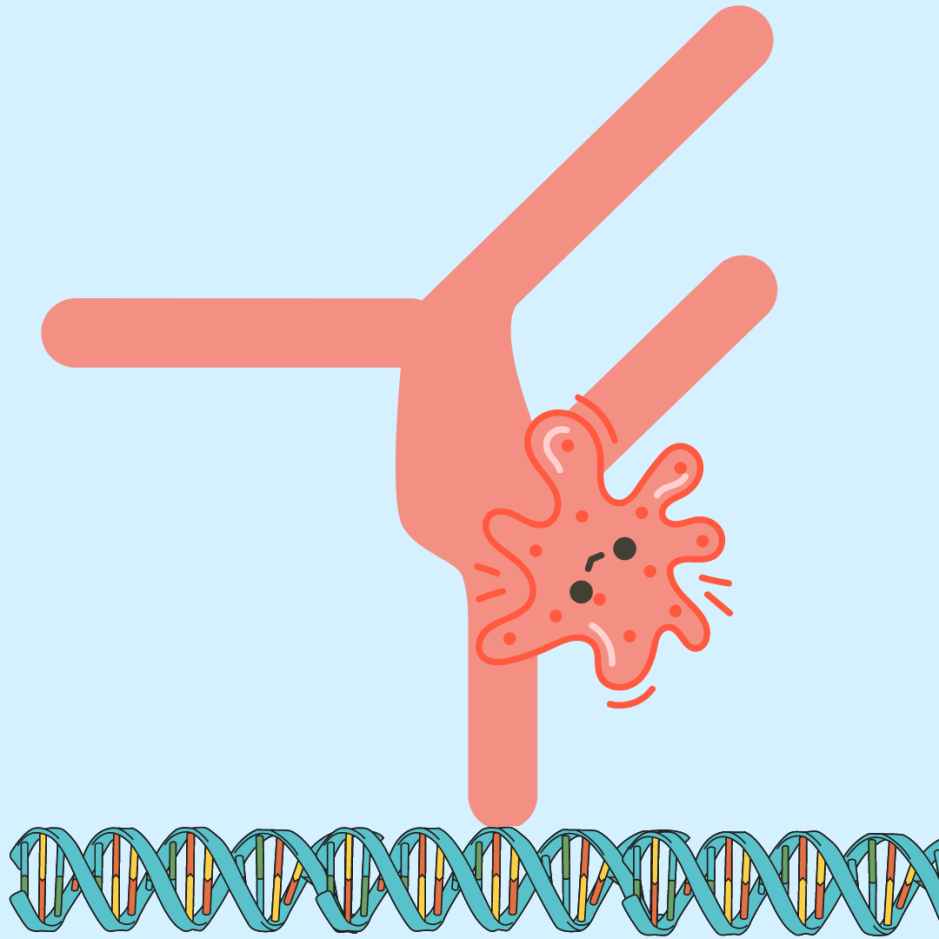
Recent research, however, has shown that using gene editing techniques can directly target the genome of the dormant virus (CRISPR/Cas9, meganucleases or similar enzymes), disrupting their function and therefore eliminating the possibility of viral reactivation and pathogenesis. So far these techniques have been developed for mice models, but they certainly are promising!
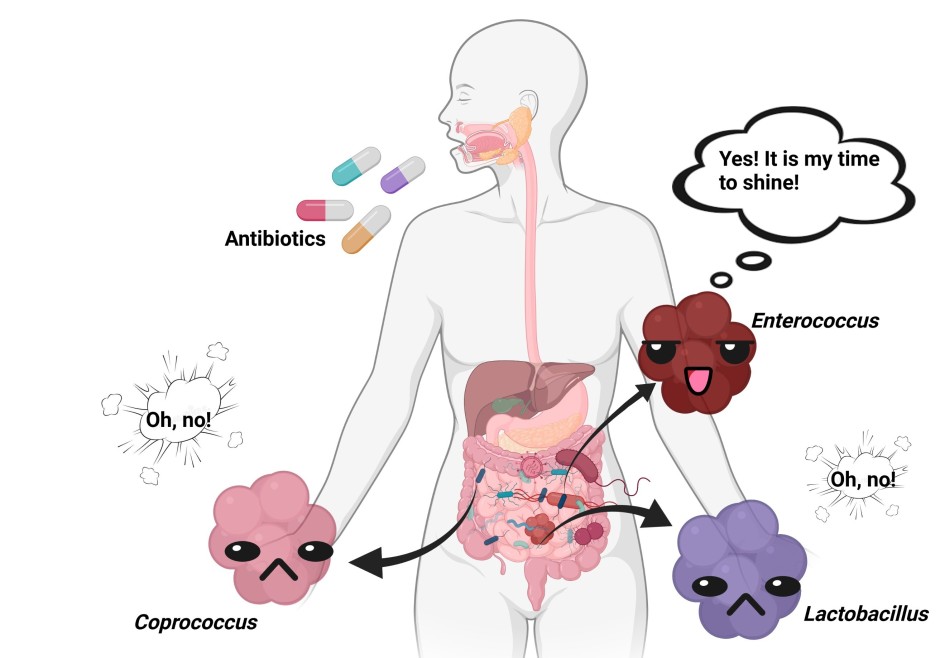
What are the genes that contribute to antibiotic resistance, and in particular vancomycin resistance in Staphylococcus aureus?
There are many ways for bacteria to resist antibiotics, depending on how the antibiotics work. Some antibiotics will inhibit cell wall synthesis (penicillin), others disrupt the integrity of the cell wall membrane (lipopeptides), inhibit protein synthesis by binding on ribosomes (chloramphenicol, tetracyclines), inhibit nucleic acid synthesis (quinolones) or inhibit metabolic pathways (sulfonamides).
The mechanisms for the antimicrobial resistance, and therefore the genes associated with them, are quite broad and can be divided in four categories:
- Pumping out the drugs (efflux pumps in bacterial membrane that actively pump out toxins)
- Limiting drug uptake (thick cell wall, hydrophobic/hydrophilic cell wall, decreased porin channels, biofilm formation)
- Modificating the target of the drug (altering binding sites for the drug so it cannot attach anymore)
- Inactivating the drug directly (degrading the drug directly, transferring a chemical group to the drug)
So any gene involved in these processes can contribute to an increased antibiotic resistance.
Drug-resistant, gram-positive bacteria such as Staphylococcus aureus are often treated by glycopeptide antibiotics. The prototypical glycopeptide vancomycin is a natural product produced by the Actinobacteria Amycolatopsis orientalis, and was discovered in the 1950s. It binds to an intermediate in the peptidoglycan layer maturation process, obstructing an important process to make a sturdy cell wall. Subsequently, this binding leads to a compromised cell envelope, resulting in osmotic stress and bursting of the cell. The work of many research groups has led to the revelation of a unique resistance mechanism, involving many enzymes that alter the basic structure of the cell envelope. In short, these enzymes change the binding structure of vancomycin on the peptidoglycan, resulting in a 1000-fold decrease in binding. There are 5 core-resistance genes involved in this: vanHAX (3 genes), vanR and vanS. These genes in combination with other Van-proteins are the biggest cause of the vancomycin resistance in Staphylococcus aureus and other dangerous gram-positive bacteria causing severe infections.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6604941/ https://pubmed.ncbi.nlm.nih.gov/31899563/
Can prebiotics feed bad bacteria in our gut? And if not, then why?
The international society association for probiotics and prebiotics’ definition of prebiotics is: “a substrate that is selectively utilized by host microorganisms conferring a health benefit”(1). Therefore prebiotics by definition should not be favouring bad bacteria in our gut. But technically yes, bad bacteria can also use prebiotics as food (2), however, prebiotics tend to make good guys flourish (such as Bifidobaterium spp. or Bacteroides spp.) limiting the growth of pathogens due to their acid production.
Does taking a probiotic actually help your intestinal microbiota? Or is it just a gimmick?
Probiotics effect have very limited clinical evidence as of today. To have a probiotic status, only one clinical trial is required (1), which limits the evidence and when several trials are done sometimes the results are contradicting. However, the international society association for probiotics and prebiotics recognizes the use of probiotics for these conditions (2):
- Helping reduce the incidence and duration of antibiotic-associated diarrhea
- Helping manage digestive discomfort (including in irritable bowel syndrome)
- Helping reduce colic symptoms in breastfed babies and occurrence of atopic issues such as eczema in infants
- Helping reduce necrotizing enterocolitis in preterm infants (3)
- Helping reduce symptoms of lactose maldigestion
- Treating acute pediatric infectious diarrhea
- Decreasing the risk or duration of upper respiratory tract infections (such as the common cold) or gut infections
Are there any benefits to supplementing postbiotics?
Postbiotics is “a composite of ‘biotic’, defined as “relating to or resulting from living organisms”, and ‘post’, a prefix meaning ‘after’. Together these terms suggest ‘after life’; that is, non-living organisms”. One proposed definition is “preparation of inanimate microorganisms and/or their components that confers a health benefit on the host”.
Postbiotics are still very new, very few clinical trials have been done as of today. However, the studies indicate health benefits for the following types of conditions:
- eradication of Helicobacter pylori infection
- reduction of symptoms in patients with irritable bowel syndrome (IBS)
- reduction of symptoms in patients with chronic unexplained diarrhoea
- abrogation of the negative effects of stress
Many more clinical trials are underway!
Salminen, S., Collado, M.C., Endo, A. et al. The International Scientific Association of Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of postbiotics. Nat Rev Gastroenterol Hepatol (2021). https://doi.org/10.1038/s41575-021-00440-6
What is the difference in gut bacteria found in people diagnosed with ADHD compared to people without? What are the latest modifications found to improve gut bacteria for ADHD?
Several studies have explored the brain-gut axis, the bi-directional communication systems between the brain and the GI track, to explore the role of the human gastro-intestinal microbiome in a variety of neurological diseases. Attention deficit hyperactivity disorder (ADHD) is no exception.
The DSM- V’s definition of ADHD (2) is quite broad as the disease itself has variable presentations within the diagnosed. Moreover, not a single factor, genetic or environmental is directly responsible for the development of the disorder. This is compounded the current hypothesis that environmental influences from pre-birth to early childhood contribute to ADHD. Subsequently, studies tend to examine those diagnosed with ADHD according to their age groups (1). However, differences in techniques, cohorts, and accountability of ADHD medication have made it difficult to have a clear comparison between ADHD and non-ADHD diagnosed individuals.
Below is the summary of different bacteria that have appeared in literature that distinguish between non-ADHD and ADHD diagnosed cohorts.
- Dialister spp. – Lattimer et al found an increased levels of Dialister spp seen in toddlers who are diagnosed with ADHD (3). This apparently contributes to increase in pro-inflammatory metabolites along the gut-brain axis. in addition to inflammation of the mind, this also contribute to decrease in pleasure.
- Faecalibacterium – Commonly noted when comparing microbiome profiles disease and healthy cohorts is Faecalibacterium. Its absence typically spells disaster. In 51 newly diagnosed ADHD children, Jiang et al. group observed a significant decrease in Faecalibacterium, a genus of bacteria important in anti-inflammatory properties (4).
- Bacteroides – In adolescents, a significant increase in Bacteroides was found in two different studies though they disagreed on other diversity and compositional parameters (Prehn-Kristensen et al. and Wang et al.)
- Bifidobacterium– The genus plays an important role in protecting the integrity of the gastrointestinal tract. In the context of the brain-gut axis it is known to code for an enzyme used in pathways that synthesize a precursor of dopamine, possibly influencing the brain via the gut-brain axis.However, studies regarding its role are in inconsistent across different age groups. In young adults, Aarts et al., observed significantly increased levels of Bifidobacterium in ADHD participants. Contrastingly, in infants between the ages of 3-6 months that later developed ADHD in childhood, there was a significant decrease in Bifidobacterium.
Ultimately, the research community is convinced that the gut microbiome plays an important role in the development of ADHD. Therefore, an exploration of therapeutic alternatives to medication are explored.
Researchers suggest that making up the deficit in important microbe-derived metabolites to improve the ADHD symptoms. For example, omega-3 Polyunsaturated Fatty Acids (omega-3 PUFA) play an important in anti-inflammatory pathways, protect the integrity of neural networks, and have a positive bi-directional influence on the microbiome gut-brain axis (8). Otherwise, treatments the microbiome to mitigate the effects of ADHD are unknown.
References:
- Bull-Larsen S, Mohajeri MH. The potential influence of the bacterial microbiome on the development and progression of ADHD. Nutrients. 2019 Nov;11(11):2805.
- American Psychiatric Association . Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Publishing; Arlington, VA, USA: 2013
- Martínez I, Lattimer JM, Hubach KL, Case JA, Yang J, Weber CG, Louk JA, Rose DJ, Kyureghian G, Peterson DA, Haub MD. Gut microbiome composition is linked to whole grain-induced immunological improvements. The ISME journal. 2013 Feb;7(2):269-80.
- Jiang HY, Zhou YY, Zhou GL, Li YC, Yuan J, Li XH, Ruan B. Gut microbiota profiles in treatment-naïve children with attention deficit hyperactivity disorder. Behavioural brain research. 2018 Jul 16;347:408-13.
- Prehn-Kristensen A, Zimmermann A, Tittmann L, Lieb W, Schreiber S, Baving L, Fischer A. Reduced microbiome alpha diversity in young patients with ADHD. PLoS One. 2018 Jul 12;13(7):e0200728.
- Wang LJ, Yang CY, Chou WJ, Lee MJ, Chou MC, Kuo HC, Yeh YM, Lee SY, Huang LH, Li SC. Gut microbiota and dietary patterns in children with attention-deficit/hyperactivity disorder. European child & adolescent psychiatry. 2020 Mar;29(3):287-97.
- Buske-Kirschbaum A, Schmitt J, Plessow F, Romanos M, Weidinger S, Roessner V. Psychoendocrine and psychoneuroimmunological mechanisms in the comorbidity of atopic eczema and attention deficit/hyperactivity disorder.
- Checa-Ros A, Jeréz-Calero A, Molina-Carballo A, Campoy C, Muñoz-Hoyos A. Current Evidence on the Role of the Gut Microbiome in ADHD Pathophysiology and Therapeutic Implications. Nutrients. 2021 Jan;13(1):249.
- Dervola KS, Roberg BA, Wøien G, Bogen IL, Sandvik TH, Sagvolden T, Drevon CA, Johansen EB, Walaas SI Behav Brain Funct. 2012 Dec 10; 8():56.
What do we know about the microbial composition of different layers of tissue in the gut and its composition’s function?
On the surface of all tissue layers exposed to the outside environment is a coating of mucin layers which is produced by goblet cells along the epithelium. These mucosal layers differ in their exact composition, but their primary objectives are to protect the tissues from invading pathogens. Commensal or “cooperative” bacteria are essential in maintaining the integrity of the mucosa. They prevent other more sinister bacteria from burrowing into the mucin layers and in turn can use the outermost layer as a nutrient source. Subsequently, the byproducts, such as short chain fatty acids of commensal bacteria utilizing mucins for energy also benefits the host.
The degree of thickness of the mucosa layer proportional to the concentration of bacteria. Within the upper gastrointestinal tract, which was previously thought to be sterile, has a thinner coating of mucins that are resistant to fluctuating acidity (MUC5AC). However, as one progresses along the gastrointestinal tract, the thickness, and the composition of the mucin (MUC2) changes.
Within the colon there are two layers of mucins, the inner and the outer layer. The inner mucin layer is primarily sterile thanks to its peptidoglycan net. If any invading pathogens were to breach this layer it could have drastic inflammatory response which could begin an unfortunate cycle of inflammation. Fortunately, a very thick outer layer of mucin and the hordes of commensal bacteria make it difficult for invading pathogens to pass. The figure below shows the outer and inner mucin layers of colon.

References:
Hansson GC. Mucins and the microbiome. Annual review of biochemistry. 2020 Jun 20;89:769-93.
How does the microbiome relate to asthma?
Asthma or airflow obstruction due to immune malfunction is not only susceptible to genetics and environmental triggers but is also influenced by both the respiratory and gastrointestinal (GI) microbiome. This is fascinating as each system has their own localized mucosal immune system (MALT) and their own bacterial communities. Though the exact nature of the relationship is not clear, researchers suggest that the cells that coat the lining of both the respiratory and GI tracts react similarly when one or the other is damaged (1, 2).
The gut lung axis, first established in 2015, demonstrates that subtle changes in the gastrointestinal tract correlates with severe changes in respiratory disease status (3). In the case of asthma, short chain fatty acids and other microbial derived metabolites, appear to be the bigger drivers of developing symptoms (3). This is supported by alterations to a development GI microbiome could have severe consequences on the long-term respiratory health of an individual. Additionally, childhood and perinatal exposure to antibiotics, factors that influence infant microbiome, and another significant disruption to a development of a healthy immune system can have long term consequences (4).
References:
- Perrone EE, Jung E, Breed E, Dominguez JA, Liang Z, Clark AT, Dunne WM, Burd EM, Coopersmith CM. Mechanisms of methicillin-resistant Staphylococcus aureus pneumonia-induced intestinal epithelial apoptosis. Shock (Augusta, Ga.). 2012 Jul;38(1):68.
- Coopersmith CM, Stromberg PE, Davis CG, Dunne WM, Amiot DM, Karl IE, Hotchkiss RS, Buchman TG. Sepsis from Pseudomonas aeruginosa pneumonia decreases intestinal proliferation and induces gut epithelial cell cycle arrest. Critical care medicine. 2003 Jun 1;31(6):1630-7.
- Barcik W, Boutin RC, Sokolowska M, Finlay BB. The role of lung and gut microbiota in the pathology of asthma. Immunity. 2020 Feb 18;52(2):241-55.
- Taylor SL, Simpson JL, Rogers GB. The influence of early-life microbial exposures on long-term respiratory health. Paediatric Respiratory Reviews. 2021 May 26.
What are the most favourable bacteria to have in your oral microbiome, and how do you cultivate a healthy oral microbiome?
The mouth is not a homogeneous environment for the resident microbiota, but offers several distinct habitats for microbial colonisation, such as teeth, gingival sulcus, attached gingiva, tongue, cheek, lip, and hard and soft palate. These oral habitats form a highly heterogeneous ecological system and support the growth of significantly different microbial communities. The teeth are the only natural non-shedding surfaces in the human body and provide unique opportunities for extensive biofilm formation, and a secure haven for microbial persistence. Early colonizers of the tooth are mainly health-associated streptococcal species, such as S. sanguinis and S. gordonii, as well as other closely related taxa. Subsequent colonization of the teeth with pathogenic species, in combination with a constant supply of carbohydrates (sugars), can lead to dental decay. There are a few host- and microbe-derived factors that can contribute to a healthy oral microbiome.
- Reduction of dental plaque by removing it (brushing your teeth).
- Fluoride: Prevents and treats caries by promoting remineralization of tooth enamel. It can also impair bacterial metabolism.
- Reduction in amount and frequency of consumption of sucrose and acidic drinks (even the ones that are sugar-free).
- Agents reducing acid production or promoting alkali generation within dental plaque (for instance arginine).
- Increasing salivary flow. Saliva has the capability to assist in buffering acids, supplying antimicrobial peptides and antibodies. Chewing gums containing polyols, such as xylitol, provide salivary stimulus without fermentable carbohydrates.
- Probiotics: Adding non-pathogenic species to outcompete the pathogenic species. Streptococcus dentisani and Streptococcus A12 are two recently described species which show particular promise as potential probiotics.
- (Specifically targeted) antimicrobial peptides: Targeted method to remove specific pathogenic species such as S. mutans, while leaving commensal streptococci unharmed.
https://www.frontiersin.org/articles/10.3389/fmicb.2018.03323/full https://www.nature.com/articles/sj.bdj.2016.865