Breaking down the microbiology world one bite at a time
Recording the gut with bacterial cameras
The power of the gut microbiome
The human gut is home to millions of thriving microbes, including bacteria, fungi, and viruses, as well as the genes they produce. Together, it is a powerful team that protects us from pathogens, builds our immune system, and converts food into energy. The composition of microbes within the gut is largely influenced by external factors, such as diet, medications, and environmental exposures. In turn, these differences in microbial composition have a large impact on disease susceptibility. For example, diabetes, obesity, cardiovascular disease, and allergies have all been linked to changes in the microbiome. While the gut is clearly a central player in human health, there are few tools to noninvasively and directly monitor this influential community of microbes.
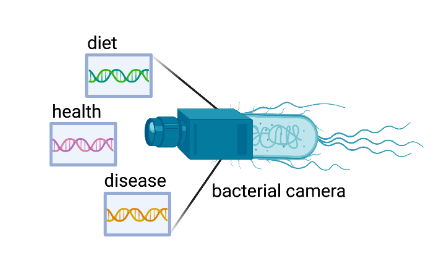
Researchers in the labs of Randall Platt (ETH Zurich) and Andrew Macpherson (Bern University Hospital, University of Bern) are trying to overcome this limitation by engineering bacterial cells that can noninvasively record changes and interactions within the gut to provide real time analyses on diet, health, and disease (Figure 1). This technology has the potential to revolutionize personalized health care, where recordings of a patient’s gut could guide disease diagnosis and treatment.

Engineering bacterial cameras
To develop this technology, the scientists equipped E. coli sentinel cells with “recording devices” that can document the gut environment. They chose E. coli because it is a naturally occurring intestinal microbe, and is regarded as a safe option for probiotic use in humans. To give these bacteria recording-abilities, researchers turned to the established CRISPR-Cas mechanism. CRISPR-Cas works as a “camera” by allowing microorganisms to capture snippets of viral RNA or DNA and store them in a region of their own genome, known as the CRISPR array. This camera enables bacteria to “remember” viruses they’ve encountered so they can more easily fight off future viral attacks and gain immunity.
In this study, instead of “remembering” viruses, the scientists altered the camera mechanism so that the bacteria can remember specific genes produced during their journey through the microbiome. In this new mechanism, the bacteria now store snippets of their own mRNA in the CRISPR array. These mRNA snapshots serve as a blueprint for which genes are being produced and used to execute cellular function. Since gene expression is largely environmentally dependent, these snapshots can provide abundant insight on interactions and changes in the gut overtime.
Recording information about diet and disease
The engineered bacteria were then used to track and report on the gut environments of living mice. To perform these experiments, the researchers orally administered the E. coli bacterial cameras to the mice, where they passed through the intestinal tract. The E. coli cells were isolated from fecal samples, and high-throughput sequencing methods were used to decipher the genetic information stored during passage through the gut (Figure 2).

The bacterial cameras were able to capture variances in gene expression in response to different nutrient supplies. Upon analyzing the recorded genetic information, the scientists were able to effectively distinguish between two different diets fed to the mice. Additionally, the gene snapshots were able to differentiate between healthy and inflamed intestinal environments, highlighting the potential of this technology to reveal early signs of disease and monitor gut health.
The future of gut health
This technology represents an effective and non-invasive method to record and report changes in the gut, providing valuable insights on diet and health. As a result, this tool can be used to aid in the diagnosis of malnutrition and disease, as well as guide and monitor nutritional and therapeutic treatments. With a proof-of-concept study successfully completed in mice, the research team aims to translate this technology to humans in the future. Extensive safety studies are currently underway to assess this possibility. Stay tuned – these bacterial cameras may soon be going live!
Link to the original post: Schmidt, F.; Zimmermann, J.; Tanna, T.; Farouni, R.; Conway, T.; Macpherson, A. J.; Platt, R. J. Noninvasive Assessment of Gut Function Using Transcriptional Recording Sentinel Cells. Science 2022, 376. https://doi.org/10.1126/science.abm6038
Additional sources:
- Schmidt, F.; Platt, R. J. Applications of CRISPR-Cas for Synthetic Biology and Genetic Recording. Curr. Opin. Syst. Biology 2017, 5, 9–15. https://doi.org/10.1016/j.coisb.2017.05.008
- https://www.genengnews.com/topics/translational-medicine/bacterial-sentinel-cells-engineered-to-record-and-report-on-gut-health/
- https://www.niehs.nih.gov/health/topics/science/microbiome/index.cfm#:~:text=The%20microbiome%20is%20the%20collection,to%20human%20health%20and%20wellness
Featured image: made by the author using BioRender.com