
Breaking down the microbiology world one bite at a time
Space Wars: The Phage Strikes Back
When we imagine the challenges of space travel, our minds usually jump to floating astronauts, cosmic radiation, or the difficulty of growing fresh lettuce in orbit. But far above Earth, inside the International Space Station, a real-life interstellar space war was taking place!
In a recent study, scientists sent bacteria and the viruses that infect them, called phages, into space to see how they behave and evolve beyond Earth’s gravitational pull. You might wonder: Why send germs to space? The answer goes beyond protecting astronauts. This research may also help scientists develop new medical tools on Earth, particularly in the growing field of phage therapy, where viruses are used to treat antibiotic-resistant infections. How does the way that viruses infect bacteria change in space? And can these new ways of infection help us fight antibiotic-resistant bacteria?
When gravity disappears, biology changes
Microgravity dramatically alters how particles move, how cells grow, and how molecules mix. On Earth, warm liquids circulate naturally due to gravity. This movement helps phages bump into bacteria. In microgravity, this ‘mixing’ largely disappears and at the same time, bacteria themselves behave differently in space. Their physiology changes, including the composition of surface molecules that phages must recognize to infect them. These changes can, starting with the first encounter, influence every step of the infection process.
Launching the experiment
The researchers chose a well‑studied microbial duo: Phage T7 and Escherichia coli BL21, a harmless laboratory strain. They prepared 32 tiny cryovials containing either phages, bacteria, or mixtures of both, then froze them solid. Half of these vials were launched to the ISS; the other half stayed on Earth as controls. All samples followed the same sequence: freeze → thaw → incubate at 37°C → refreeze. After their orbital adventure, the vials were returned to Earth, thawed one final time, and analyzed to see how the microorganisms within had fared.
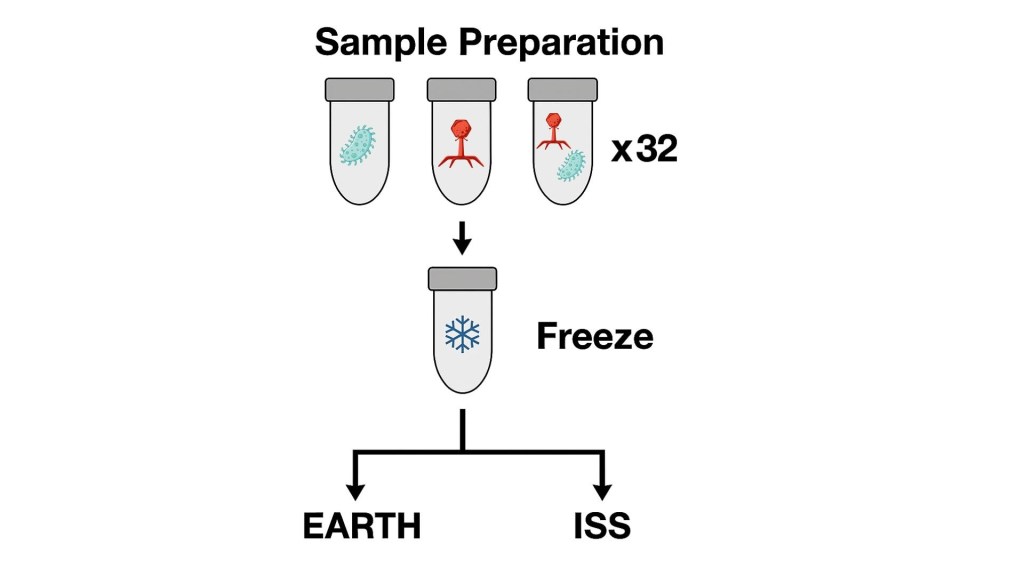
There were two main experimental timelines. The first timeline included short-term incubations of 1, 2 or 4 hours to test how quickly phage infection begins in microgravity. The second timeline existed out of a long-term 23-day incubation which allowed for many rounds of replication, and therefore evolution, to occur. After recovery, the team measured bacterial and phage abundance, sequenced their genomes to detect new mutations, and analyzed mutational variants to see how microgravity shaped phage evolution.
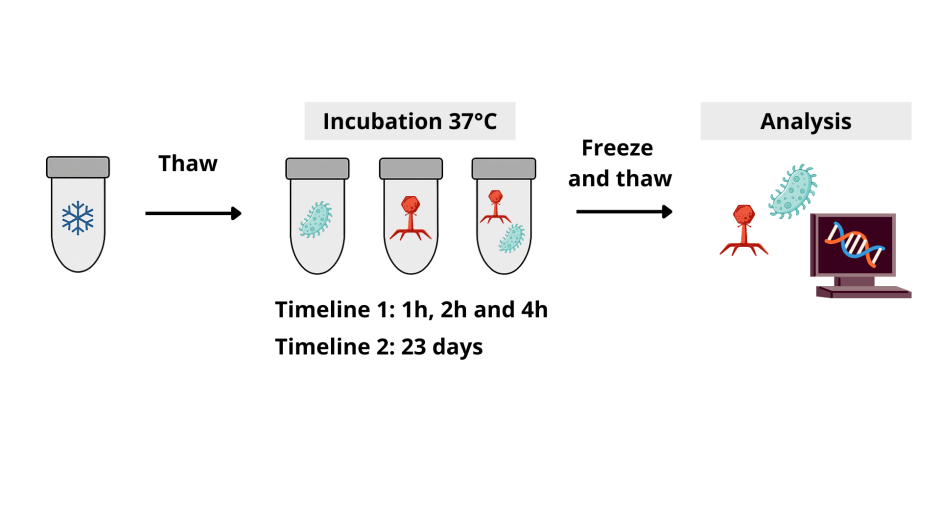
Microgravity slows the battle
On Earth, T7 usually infects and destroys E. coli within 20–30 minutes. The earthbound controls behaved similarly (with some slowing due to freeze–thaw effects), but in microgravity? For the first four hours, nothing happened. No rise in phage numbers. No fall in bacterial numbers. The phages simply weren’t encountering their hosts. But by the 23‑day mark, things had turned around: phage numbers had surged, confirming that phage infection does eventually succeed even if space dramatically slows the clock. The researchers speculated that this delay was caused by two combined effects in microgravity: 1) Lack of fluid mixing: Without convection, phages drift slowly and may rarely collide with bacteria, and 2) Altered bacterial physiology: Changes in membrane structure or receptor expression can make infection less efficient. To test these hypotheses, the researchers continued their experiments.
The evolutionary space race
To understand the impact of space on membrane structure and receptor expression, the researchers looked at the genomes of the phages and bacteria. After 23 days, whole-genome sequencing revealed a wealth of new mutations in both species: Phages accumulated mutations across many genes, but the strongest changes appeared in proteins helping the phage attach to or interact with the bacterial surface. Their mutation patterns suggest that phages had to adapt to changes in the bacterial outer membrane that occur under microgravity. Bacteria, in turn, evolved mutations in genes involved in outer membrane structure, stress response and metabolism. These changes likely helped bacteria resist phage infection either by modifying their surface receptors or by adjusting their growth strategies. These mutational patterns reveal a true microgravity‑specific evolutionary arms race between phages and their bacterial hosts.
Mutations that change the game
And this was not the only discovery suggesting that the bacterial surface itself changes in microgravity. One of the most surprising findings came from the deep mutational scanning (DMS) library. DMS is a technique where scientists create thousands of versions of a protein, each containing a single mutation. They then test them all at once to see which mutations help or hurt function. It’s like running evolution on fast‑forward and watching which variants thrive. In this experiment, 1,660 variants of the phage’s receptor binding protein (RBP) tip, its grappling hook with which it latches to bacteria, were tested at the same time.
The mutations that performed best in space were totally different from those favored on Earth. Space‑selected variants favored hydrophobic, internally buried changes whereas earth‑selected variants favored positively charged, outward‑facing residues that normally interact with bacterial receptors. This strongly suggests that the bacterial surface itself changes in microgravity, forcing phages to evolve new strategies for attachment.
Space-trained phages
To see if these space‑selected insights had practical value, the researchers tested two groups of receptors: one combining the best mutations selected in microgravity and the other combining the best mutations selected on Earth. They then tested these groups on two E. coli strains that are normally resistant to T7. The researchers found that the microgravity‑selected variants outperformed the ones found on Earth This shows that space revealed beneficial mutations that Earth-based evolution simply did not uncover.
Coming back down to Earth
This study shows that microgravity exposes microorganisms to entirely new selective pressures. Pressures strong enough to reveal genetic solutions that may never emerge on Earth.This could transform phage engineering by uncovering new parts of viral proteins that influence how they infect bacteria. This may help us design phages that infect antibiotic‑resistant bacteria.
Link to the original post: Microgravity reshapes bacteriophage–host coevolution aboard the International Space StationHuss P, Chitboonthavisuk C, Meger A, Nishikawa K, Oates RP, et al. (2026) Microgravity reshapes bacteriophage–host coevolution aboard the International Space Station. PLOS Biology 24(1): e3003568. https://doi.org/10.1371/journal.pbio.3003568
Featured image: Image created using M365 Copilot.















