Breaking down the microbiology world one bite at a time
Wolbachia hacks the mating game
Inside the cells of many insects lives Wolbachia, a microscopic bacterium only ~1 μm across. Despite its tiny scale, it’s incredibly widespread, infecting over 66% of insect species, including flies and mosquitoes. Bacteria like this are called endosymbionts (from Greek: endo = inside, symbiont = living together), because they reside within another organism. But Wolbachia is far from a passive resident. Its presence can reshape host biology in two main ways: by triggering immune system responses and by releasing bacterial proteins that interfere with cellular processes. Since Wolbachia is maternally transmitted, any effects that boost female reproductive output can enhance its spread. Since Wolbachia is maternally transmitted, it must infect the germline to be passed to offspring. However, its ability to alter host behavior is expected to act through the brain.
Understanding how Wolbachia manipulates its hosts can reveal how microbes influence animal behavior more broadly. Beyond the lab, this knowledge has real-world applications: Wolbachia is already being used to control mosquito populations and reduce the spread of viruses like dengue and Zika, both by limiting the viruses mosquitoes carry and by disrupting their reproduction. Uncovering the mechanisms behind its spread could help improve these control strategies.
In a study by Warecki et al., researchers asked a simple but intriguing question: Does Wolbachia infection change female mating behavior? To answer this, they turned to the fruit fly Drosophila melanogaster, a powerful model system with well-characterized genetics and easy laboratory handling.
Flies, Vials, and Mating Trials
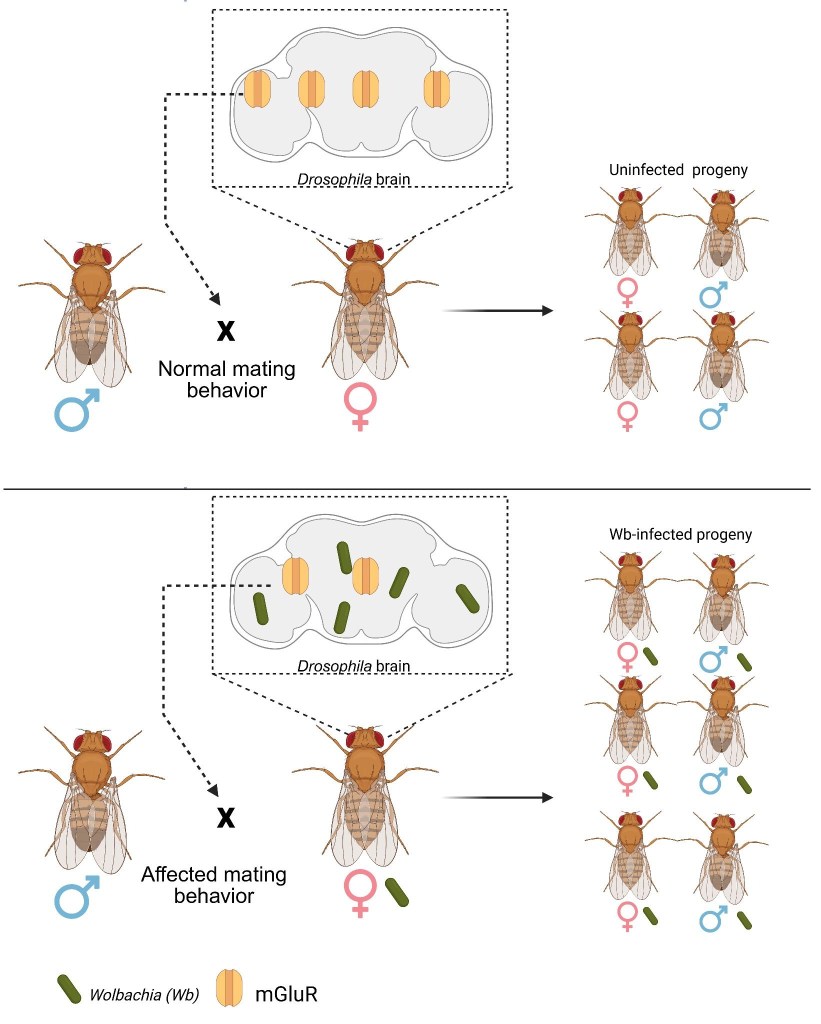
To find out, the researchers compared infected and uninfected female flies across several experiments. Drosophila can be easily maintained in small food-filled vials where females lay their eggs. Like this, researchers could select virgin females, perform controlled crosses, and track the number of offspring produced. First, they performed mating assays using males from the same species. Second, they looked at mating between closely related species (hybrid crosses). In both cases, infected females produced more offspring. But why? Looking closer, they found that infected females were more receptive to mating, they mated more often, even in situations (like cross-species mating) where they would normally be more selective. In other words, Wolbachia increases female mating frequency, even in situations where mating would normally be less likely.
What’s in her mind?
Next, the researchers investigated what was happening in the brain. Using advanced imaging, they found Wolbachia localized in regions linked to sensory perception and mating behavior, hinting at a direct neurological effect. Protein analysis revealed even more. Infection altered the levels of many host proteins. One standout was mGluR (a glutamate receptor), which was less abundant in infected brains. When researchers artificially reduced mGluR in uninfected flies, those flies behaved just like infected ones, showing increased mating success. Why does this matter? mGluR normally helps dampen neural signaling. With less of it, neurons may become more excitable, potentially making females more responsive to mating cues. The researchers found other proteins that changed in abundance, suggesting a coordinated shift in brain signaling pathways. Interestingly, the team also detected many Wolbachia-derived proteins in the brain, some of which may directly interact with host proteins—pointing to a more hands-on manipulation by the bacterium.
A Happy Ending for Wolbachia?
Taken together, these findings suggest that infected females mate more and produce more offspring. Because Wolbachia is transmitted through eggs, a subtle shift in behavior can enhance its spread, turning increased receptivity into a powerful evolutionary advantage. A tiny bacterium, quietly rewiring the brain, just enough to tip the scales in its favor.
Link to the original post: Warecki, B., Vega, G., Fowler, S., Hartzog, G., Karr, T. L., & Sullivan, W. (2025). Wolbachia-mediated reduction in the glutamate receptor mGluR promotes female promiscuity and bacterial spread. Cell reports, 44(5), 115629.
Featured image: Image generated with Google Gemini.