
Breaking down the microbiology world one bite at a time
How Testosterone Tips the Balance of Infection
We are more bacteria than human by cell count. Our skin alone is home to billions of microbes, including thousands of different species of bacteria. Many of these microbes are beneficial, helping maintain the skin’s protective barrier and defending against harmful pathogens. But among them lies a well-known culprit behind many skin infections: Staphylococcus aureus.
You may recognize S. aureus from MRSA (methicillin-resistant Staphylococcus aureus), a strain that is notoriously difficult to treat because of its resistance to many antibiotics. But did you know that even non-resistant strains can be dangerous? Under certain environmental conditions, S. aureus shifts from a quiet resident of the skin to an aggressive pathogen, capable of causing painful infections.
Not all skin microbiomes are created equally, and neither are the conditions that shape them. A recent study from researchers at the University of San Diego found that the severity of S. aureus skin infections increases in individuals with higher levels of testosterone. On average, male-identifying participants secreted more testosterone at the skin surface than female-identifying participants. In correlation, males also had higher levels of S. aureus skin colonization and more severe infections.
These findings were mirrored in animal models: male mice produced more testosterone and developed more S. aureus-induced skin damage compared to female mice. Symptoms included greater tissue necrosis (more skin cell death) and worse overall disease scores.
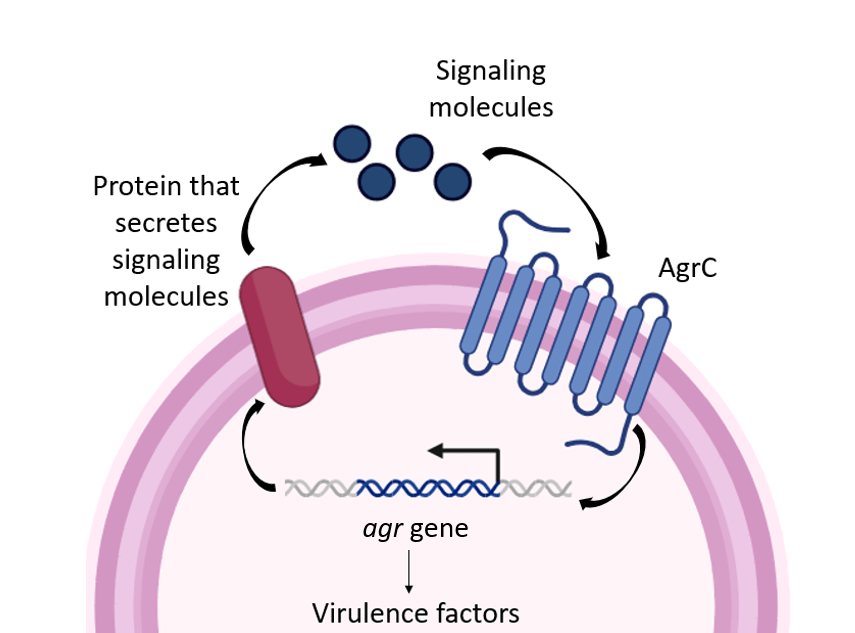
The underlying difference can be attributed to how bacteria “talk” to each other. S. aureus uses a communication system known as quorum sensing, in which bacterial cells release and detect small signaling molecules to monitor their population density. Once enough bacteria are present, these signals trigger coordinated responses, including the activation of genes involved in virulence and toxin production.
One of the key quorum sensing systems in S. aureus is the accessory gene regulator (Agr) pathway. This pathway acts as a molecular switch between harmless colonization and active infection. In this study, researchers demonstrated that testosterone can interfere with this system in a surprising way.
Specifically, testosterone binds to a receptor protein called AgrC, which ordinarily detects bacterial signaling molecules. In doing so, testosterone effectively “switches on” the Agr pathway, activating virulence even in the absence of the bacteria’s usual communication signals. A host-derived hormone becomes a false signal, nudging S. aureus to move toward a more aggressive, infectious state.
These results were validated using a clever molecular twist: the enantiomer of testosterone. An enantiomer is a mirror-image version of a molecule. While it looks nearly identical, its flipped structure prevents it from binding to the same targets in the same way. In this case, the enantiomer was unable to activate AgrC and instead blocked the interaction, preventing the downstream activation of virulence pathways. As a result, damage to the skin of the host was reduced.
These findings point toward an intriguing therapeutic possibility: instead of killing bacteria outright, can we disarm them by disrupting their communication systems? Traditional antibiotics often drive resistance, but anti-virulence strategies (like blocking quorum sensing) take a different approach. By preventing bacteria from switching into a harmful state, they may reduce disease severity without applying the same selective pressure that promotes antimicrobial resistance. Such strategies are already being explored. For example, probiotics containing bacterial strains that produce molecules that interfere with quorum sensing are currently in development as potential S. aureus therapies.
As it turns out, host-microbe interactions are more of a complex conversation than we previously thought. Hormones like testosterone, long studied for their roles within the human body, may also act as signals that microbes can intercept and reinterpret. In this case, a molecule intended to regulate our own physiology becomes, inadvertently, a cue for bacterial attack.
Other interesting questions to consider going forward include: How do our bodies shape risk of infection, and can individuals have different levels of susceptibility to infections based on their hormonal profile? How widespread is this phenomenon across other microbes? Continuing to understand these hidden signals could reshape how we prevent infection altogether.
Link to the original post: John, M.S.; Chinnappan, M.; Sturges, C.I.; et al. Skin androgens regulate Staphylococcus aureus pathogenicity via quorum sensing. Nat. Microbiol. (2026). 11: 704-717.
Additional sources:
- To read about a probiotic that disrupts S. aureus quorum sensing, click here.
- For more information on quorum sensing, click here.
Featured image: Image created by author using stock images and assembled in PowerPoint.















