
Breaking down the microbiology world one bite at a time
Computers vs. pathogens: the final frontier?
Antibiotic resistance is a huge problem. Rapidly, our repertoire of antibiotics used to treat serious bacterial infections is becoming obsolete. To give you some perspective, in 1928 Alexander Fleming discovered the antibiotic penicillin (1), which changed medicine forever (well, at least up until now, around 70 years later). This antibiotic was used in the trenches of WW2 to help treat infection (2). By the time penicillin became commercially available in the early 1940’s, various bacteria had already begun to acquire resistance to this “miracle drug”.
Bacteria can acquire resistance to penicillin and other antibiotics in several ways, including sharing genetic information. Though never entirely harmless, some bacteria that acquire resistance are more consequential than others, such as my favourite bacterium Pseudomonas aeruginosa. Given the concerning rise in the acquisition of antibiotic resistance, we need new tools to tackle this concerning, global issue.
What is a “Pseudo…” what did you call it?
Pseudomonas aeruginosa is a human pathogen that preferentially infects people with upper respiratory tract diseases, such as Cystic fibrosis (CF) (3). Also of great concern is that P. aeruginosa can also infect patients through medical equipment. This is achieved through forming biofilms on catheters and other implants. This makes patients prime targets for infection. Throughout my time in research, I was fascinated by the immunology behind the preferential infection, which drove me to learn more.

The immune system in CF patients does not function to the same level as yours or mine, meaning that the white blood cells normally tasked with removing pathogens – such as P. aeruginosa – are not very effective (3). This allows the pathogen to colonize and form biofilms, propagating the infection. These biofilms are a collection of bacteria surrounded by a protein and sugar matrix – impenetrable to conventional antibiotics. If you watch Star Trek, you can think of a biofilm as the “Shields” of the USS Enterprise.
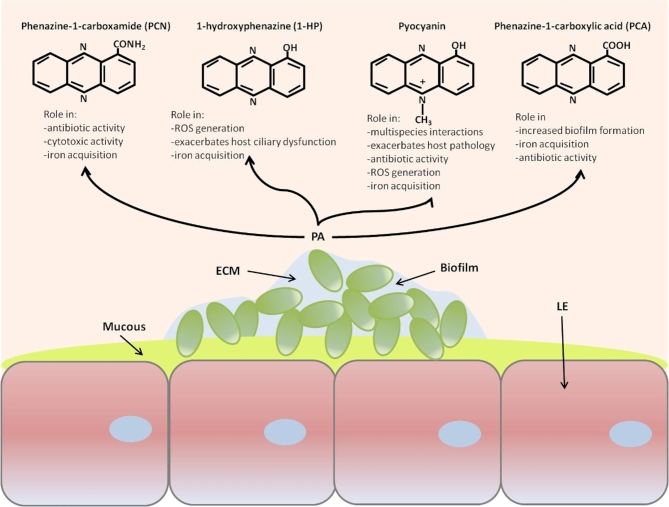
Another tool P. aeruginosa has in its infection repertoire are phenazines – molecules that scavenge iron and other nutrients for its host (4). Because of this scavenging ability, among other functions, phenazines, directly and indirectly, kill other bacteria in the vicinity (essentially taking a MicroBite out of the competition – sorry I had to). Following our metaphor, the phenazines could be considered as the “Phasers” of the Enterprise.
Turns out that both biofilms and phenazines are closely linked – for biofilms to form, nutrients need to be supplied to the bacteria. But to get these nutrients, the bacteria need to produce phenazines. As an interesting note, while phenazines are produced all the time by the bacterium, phenazine production is significantly enhanced during biofilm formation. So, could phenazines be the target of therapeutics? As an enticing tidbit, P. aeruginosa strains that cannot produce phenazines are more susceptible to antibiotics.
So we want to kill the Enterprise? But I like Jean-Luc.
We want to remove the shields of the Enterprise by disarming the phasers first. But then yea, I guess we are killing the Enterprise eventually…
A natural way to disarm the phasers phenazines is to degrade them using enzymes – and nature has provided us with such an enzyme – PodA (think of it as the phenazine killer). As scientists, we would typically test this enzyme by extracting it from the host organism and see the effects on its target. But in this case, PodA is not produced in sufficient quantity for experimental use (5). As such, VanDrisse and colleagues turned to computers to help with the production of PodA in a recent study. Computers are rapidly becoming a key component in the experimental pipeline in all fields of biology.
Computers can look at the structure of proteins and change them on the molecular level to be more efficient (6). What we would typically do is assess the structure of the protein, and add enhancements to increase folding efficiency or reduced susceptibility for degradation. In fact (going on a slight tangent), if you have heard of GFP (green fluorescent protein), scientists manipulated the structure of this protein to enhance its folding ability, as well as making it impossible to degrade. Sticking with GFP, we have also made versions of this molecule that can be resistant to changes in pH (a molecule called pHluorin if you’re interested)! In this study, the scientists used computers to enhance the production of PodA (they report a 20-fold increase in production) based on the introduction of 5 mutations within PodA compared to the starting form. These mutations allow for the efficient packaging of PodA into microvesicles.
Previously, the 3D packaging of PodA was inefficient for mass production. With these 5 mutations, the spatial orientation of PodA allows for it to be packaged more efficiently so more PodA can be secreted in these microvesicles, thus increasing productivity. The enhanced production of PodA allows scientists to extract more of this enzyme. They next tested the effect of PodA on phenazines And the result from those experiments confirms that PodA does indeed degrade phenazines. As per the prediction, this also leads to the abolition of Pseudomonas biofilms. So, the study was a success.
That’s cool and all, but why should I care?
In a world where bacteria are gaining resistance to antibiotics at an exponential and concerning rate, new therapeutics, and indeed new ideas to combat these bacteria, are paramount. I, for one, was infected with a multidrug resistant form of Streptococcus pyogenes years ago. I went on three antibiotics and didn’t get better. The doctors had to treat the infection with Vancomycin – essentially one of the last resort antibiotics. This was 6 years ago now, and the pathogens have not stopped adapting to antibiotics. Soon, these “miracle” drugs as they were once called, will no longer be effective. Thus, using computers to design new proteins, or alter existing ones, in the fight against human pathogens is another tool in our toolbox. Paraphrasing Captain Picard:


References:
1. Fleming, A. 1944. The Discovery of Penicillin. British Medical Bulletin, 2: 4-5.
2. Myers, W.F., Grossman, D.M., and Wisseman, C.L. 1984. Antibiotic susceptibility patterns in Rochalimaea quintana, the agent of trench fever. Antimicrobial agents and Chemotherapy. 25: 690-693.
3. Rossi, E., et al. 2021. Pseudomonas aeruginosa adaptation and evolution in patients with cystic fibrosis. Nature reviews microbiology. 19: 331-342.
4. Vilaplana, L., and Marco, M.P. 2020. Phenazines as potential biomarkers of Pseudomonas aeruginosa infections: synthesis regulation, pathogenesis and analytical methods for their detection. Analytical and Bioanalytical Chemistry. 412: 5897-5912.
5. VanDrisse, C.M., Lipsh-Sokolik, R., Khersonsky, O., Fleishman, S.J., and Newman, D.K. 2021. Computationally designed pyocyanin demethylase acts synergistically with tobramycin to kill recalcitrant Pseudomonas aeruginosa biofilms. Proceedings of the National Academy of Science. 118: e2022012118.
6. Kazmirchuk, T., et al. 2017. Designing anti-Zika virus peptides derived from predicted human-Zika virus protein-protein interactions. Computational biology and chemistry. 71: 180-187.
7. O’Brien, S., and Fothergill, J.L. 2017. The role of multispecies social interactions in shaping Pseudomonas aeruginosa pathogenicity in the cystic fibrosis lung. FEMS Microbiology Letters. 364: 1-10.
Featured image: The big model of the USS Enterprise, NCC-1701-D, hanging at the Star Trek Experience in the Hilton Hotel, Las Vegas, Nevada, by David Grant under https://creativecommons.org/licenses/by-nc/2.0/



































































































