Determining the causal link between diseases and microbiota is still a challenge in microbiota research. But animal models can help.

Breaking down the microbiology world one bite at a time
Small animals, big conclusions- pros and cons of HMA models.
Determining the causal link between chronic disease aetiology/prognosis and microbiome is challenging. Scientists have since tried a variety of methods to determine the causal link that could explain the association of microbes and disease. Enter the Human Microbiota Associated (HMA) murine models.
HMA murine models are germ-free mice (aka mice that are raised in a sterile environment) that received faecal matter from patients diagnosed with chronic diseases and/or healthy volunteers. If the disease manifests itself in the rodents after transplanting from the sick patient, then there is a basis to causality. Unfortunately, that’s all that can be said about the method as it lacks standardization. The pros and cons of the method were explored by Walter and their colleagues’ systematic review of 38 studies.
First the reviewers establish the pros of using HMA murine models:
- You will get some insight on mechanisms of pathology in rodents after receiving faecal matter while being able to control environmental factors such as housing and food.
- One can compare whether one rat’s response to microbial communities from a healthy volunteer to another rodent’s response to a donation from a sick patient.
- It’s easy to incorporate sequencing technology such as metagenome sequencing into your experiment design to readily monitor the evolution of and determine differences in gut-microbiome composition.
These models should allow us to adequately explore the causal links between the microbiome and diseases. Yet, our reviewers underline some of the limitations associated with the model and give suggestions on how to improve the model.
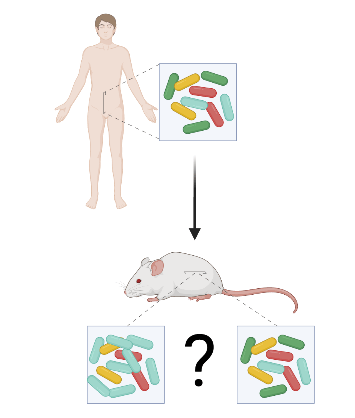
Integrity of the microbiome from donors. Microbes taken from human donors do not always successfully colonize the gastrointestinal tract of the rodent recipients. The few taxa that do colonize lay the foundation for a completely different composition. The reviewers found studies that admitted that the integrity of microbiome when taken from the human donor was damaged and yet claim that they found positive associations between the microbiome and disease. It’s important to check whether or not the microbiomes in the rodents adequately represent the human donors.
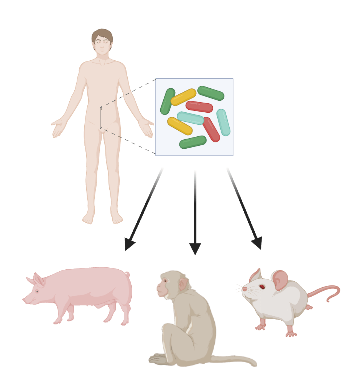
The models themselves. The gut microbiome has been shown to play an important role in development in both immune and nervous systems. Given that these rodents were raised in a sterile environment, they are susceptible to immunological and neurological problems. Moreover, rodents’ immune systems differ from humans, they are not ideal models when it comes to representing the human immune system- problematic if one were to study the link between microbes and autoimmune disorders. This problem could be addressed if rodents are humanized via incorporation of human immune cells or if other models whose immune systems better reflect humans, apes or pigs, are used.

Homogenizing heterogeneous diseases. The methods for creating and designing HMA murine model-based experiments vary greatly. Each individual’s microbiome is unique, like a fingerprint. Despite this, there are several studies that pooled the donor samples, confounding any individual features that might have been associated with disease status/severity. Therefore, it is important to ensure that there are a sufficient number of donors to accurately represent the biological variation seen in the clinic.
Numbers are everything. Another element is the statistical implication of pooling the faecal donors. When you have more rats than donors, you overrepresent your results. For example, if in reality, only a few people with disease have a significant number of bacteria A. By pooling faecal donors the results of that experiment would indicate that all people with the disease should have a significant number of Bacteria A. In reality, the most statistically accurate papers are those that have an equal number of donors and recipients.
Ultimately, the HMA murine model is not the only approach to studying causal links. Advancements in statistical models that infer causality and strides in modulating the human microbiome directly are some of the new methods to better understand the microbiome disease interface. The combination of these techniques in addition to a standardized use of HMA models can yield some interesting results in the future.
Doi: 10.1016/j.cell.2019.12.025