
Breaking down the microbiology world one bite at a time
Nature vs Nurture: Valuable lessons from a tiny worm
The philosophical question of why we do what we do is quite old. Last century, biology attempted to provide an answer by focusing on genetics. Many thought that what creatures are and do is deeply encoded within their genes. However, the field of epigenetics started to develop in 1957(1), and scientists showed that genes are regulated in many different ways. In fact, it is well known that experiences can alter genetic expression. But researchers went even further. Could the experience of an individual shape gene expression and behavior in their offspring? To answer the question, the model organism Caenorhabditis elegans has been, and still is, of high importance.
Some background on C. elegans.
This tiny nematode is a transparent worm used often in genetic and epigenetic research. It can be grown in the lab, measures about 1 mm, has about 200-300 offspring in 2-3 weeks, and feeds on bacteria(2). Despite its simplicity, it shares many essential biological pathways with more complex organisms, including humans. In its natural environment, C. elegans encounters various bacteria, some of which are infectious. Understanding how these worms respond to harmful bacteria can provide broader knowledge into fundamental biological processes.
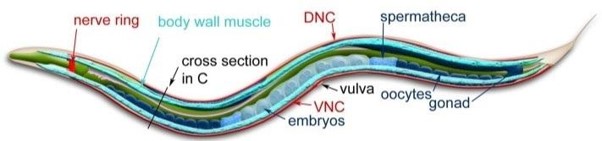
Modified from Corsi et al, 2015.
Avoidance of harmful bacteria in parents and offspring
Scientists wanted to know if C. elegans developed a preference for a food source over another. They fed some worms a traditional, non-toxic food source, and others different food sources, with different bacteria found in the wild, some which cause disease in the worms, and some which do not. After some time, they provided both food alternatives to the same worms. In most cases, the worms didn’t show a strong preference, but when they had been fed Pseudomonas vranovensis, a pathogenic bacteria, they actively avoided consuming it again. Not only that, but their direct offspring also showed an avoidance of this infectious bacteria, even when they never were in contact with it, only their parent was.
Transgenerational inheritance of behavior
Knowing the avoidance of this bacteria was passed from parent to offspring, a new question needed to be answered. Will the behavior pass to the grandchildren and even further generations? The researchers performed the same test, providing non-harmful bacteria and P. vranovensis to the following generations with grandparents exposed to this strain of bacteria. They found the avoidance of P. vranovensis perpetuated until the 4th generation. In the 5th and later generations, the worms no longer avoided the bacteria.
How is this behavior shaped and inherited?
The answer lies in the machinery within the small bacterial cells the worms consume. It has been previously discovered that C. elegans have a wide range of molecular tools to take RNA from bacteria they eat. When they eat P. vranovensis, a small RNA produced by the bacteria is identified and taken up by the worms. This small RNA is copied, and brought to the sensory neurons.
In the sensory neurons, a gene is expressed, making a messenger RNA. Normally, the cells would then use this messenger RNA to create a protein. But the bacterial RNA matches a small section of this RNA, and the worm cells then destroy the messenger RNA. And as a result, the protein is not created. In short, a small RNA from the bacteria regulates the expression of a gene in the neurons of the worms. This changes the behavior of the worm, making it avoid the harmful bacteria.
Lastly, the small RNAs from the bacteria are also passed to the germline, the cells that will form new offspring. This way, the RNAs will be present in the next generation of worms, where they can regulate gene expression in the neurons to continue avoidance of the harmful food source.
Why does inheritance stop, and broader insights into gene regulation.
The change in gene expression in the neurons of the worms triggers avoidance of the harmful bacteria, but also of other species of bacteria. Moreover, other small RNAs from other bacterial species also inhibit the same gene, triggering the same avoidance. Not all of these species are actually harmful to C. elegans, and the researchers suggest that this could be the reason for why the inheritance is not sustained after the 5th generation.
Conclusions
- Researchers have found that C. elegans learns to avoid the toxic bacteria P. vranovensis after ingesting it.
- The avoidance occurs due to small RNA in the bacteria. This RNA is taken by the worms and triggers a change in gene expression in the sensory neurons of the worm.
- The RNA also passes to the next four generations, triggering similar avoidance, but it is not inherited in further generations.
By exploring how experiences change the behavior of this tiny nematode and their offspring for several generations, scientists hope to one day understand more about how other animals and even humans are affected by their experiences at a molecular level.
Link to the original post: Ann K Corsi, Bruce Wightman, Martin Chalfie, A Transparent Window into Biology: A Primer on Caenorhabditis elegans, Genetics, Volume 200, Issue 2, 1 June 2015, Pages 387–407, https://doi.org/10.1534/genetics.115.176099
Featured image: C. elegans under a microscope. Photo by Coleen Murphy. https://www.flickr.com/photos/nihgov/30341767104















