
Breaking down the microbiology world one bite at a time
Bacterial nomads
After reading this article, you may never look at clouds of flying dust in the same way. Or at least that is what happened to me. Imagine you are standing in the middle of an arid landscape during the drought season. A strong, deceitful wind begins to blow. You instinctively cover your mouth and eyes from the dust that storms towards you. Your breathing becomes difficult. We need no explanation to understand that breathing dust is unhealthy. Yet, many of us ignore that dust is a bioaerosol, i.e., microorganisms “stuck” to inert particles (such as clay). Bioaerosols travel through our planet’s different ecosystems and impact life on Earth. Due to desertification and land-use changes, there is a projected increase in atmospheric dust loading. This generates the urgent need to understand the dust-particle microbiome, an understudied topic.
During dust storms, bacteria must somehow cling to dust particles. They survive in this harsh habitat while transported to distant, more favorable environments. A bacterial member of dust is the phylum Firmicutes, including bacteria from the genus Bacillus. Under adverse growth conditions, bacilli can activate “survival mode.” They manage this using two distinct strategies: sporulation and biofilm formation. Previous work suggested biofilm formation was crucial for the successful association of bacilli with dust. However, this was not backed up by experimental data. A fundamental aspect of biofilm formation is the generation by the bacteria of an extracellular matrix. This is a “glue” that keeps bacterial cells together and protects them from low humidity and nutrient scarcity. It also allows the bacterial community to associate with an inert surface (e.g., dust particles). Ilana Kolodkin-Gal and colleagues set out to study the relationship that exists between biofilm formation and bacterial survival in dust. First, they collected dust samples and then isolated bacilli from them. Then, they studied whether these bacteria were capable of forming biofilm when encountered with dust. For this, they used both sterile and synthetic dust particles. Figure 1 outlines the complete process for carrying out the study. The researchers collected dust from dust storms using an air-sampling machine called the Coriolis Compact sampler. The sampler processed 50 litres of air per minute, collecting dust into sterile tubes. Once in the lab, the dust samples were mixed with liquid and then heated up; this heating step allowed only spore-forming bacteria, those that can survive high temperatures by forming spores, to remain alive. This narrows the analysis to a particular group of hardy, culturable, spore-forming bacteria. At the same time, this excludes bacteria that cannot survive heat.
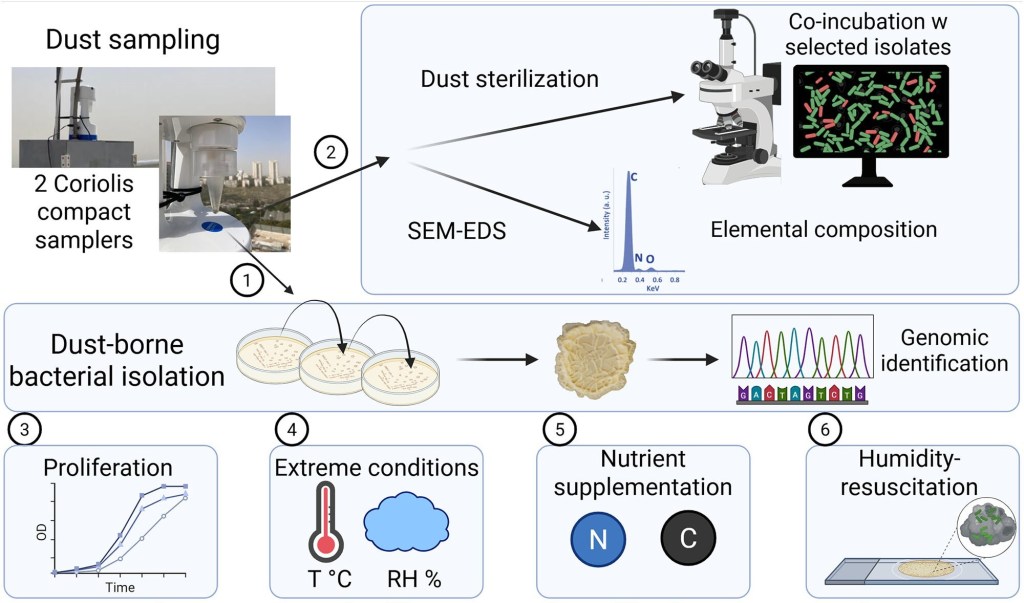
The researchers first verified that Bacilli were in fact present in the dust samples under study. They identified these bacteria using genetic and chemical tests, and together these species made up the group of microbes found in the dust storms: the dust-storm microbiota (Figure 1,2). Next, they looked at how each type of bacteria behaved in the lab. They measured two things: how well the bacteria grew and how well they formed biofilms in three different cultivation media (Figure 1,3). Because the media markedly differed in their compositions, this allowed the researchers to draw powerful conclusions concerning the nutrients the bacteria need. The great majority of the bacteria thrived in a tailored medium low on nutrients, suggesting a strong compatibility of this medium with the dust habitat. The researchers then tested the formation of complex colony biofilms across the different media, finding intricate 3D pattern formation in all isolates. Overall, the study showed that bacterial growth and biofilm formation are not the same process and are controlled by different factors.
For the next step, the team studied how each isolate could survive under extreme environmental conditions (Figure 1,4). They varied the temperature and relative humidity, using the same culture medium, and monitored colony morphology and expansion parameters of the isolates cultivated in each condition. The results showed that bacilli found in dust storms show resilience to multiple stressors. They can survive the harsh conditions of being carried through the air, including high temperatures (45°C) and very dry air. Even under these conditions, the bacteria were still able to grow and produce the sticky material that helps them form biofilms. Finally, the researchers dug deeper, monitoring the activity of the genes involved in biofilm formation and observing how the bacteria behaved at the single-cell level (Figure 1,5-6), among other experiments. Together, these tests supported the idea that forming biofilms is key to helping dust-dwelling bacteria survive. However, a key finding of this study was revealed when they evaluated growth and biofilm formation in two distinct dust environments: sterilized natural dust from dust storms, containing residual carbon from original bacterial dust-inhabitants, and artificially composed dust (composed of only the inorganic building blocks of dust). In this assay, a lab model bacterium (B. subtilis 3610) grew better when cultured in sterile dust than in synthetic dust, highlighting the importance of the organic component in dust, represented by the “carcasses” of previous inhabitants.
The scientists propose that extracellular matrix production is an adaptive trait essential for the survival of bacteria in this particular habitat. In other words, dust-inhabiting bacilli must be capable of forming biofilm to survive. Like every significant scientific discovery, this work inspires many questions. What is the origin of this dust micro-niche adaptation? And what interactions take place among dust particle bacterial communities? Future studies should also focus on the broader implications for Earth. Does long-distance bioaerosol dispersion carry associated risks for human health? Does biofilm formation contribute to global carbon sequestration processes?
There is surely more to come regarding research on the dust microbiome, especially taking into account its profound environmental impact. While science deciphers these elusive, nomadic, bacterial communities, we can only wait for the dust to settle…
Link to the original post: Lang-Yona, N., Lahav, E., Levy Barazany, H. et al. Bacillus biofilm formation and niche adaptation shape long-distance transported dust microbial community. Commun Earth Environ 6, 551 (2025).
Featured image: https://chatgpt.com/s/m_691b2d56a74c8191aa033f66bef6255e















