
Breaking down the microbiology world one bite at a time
Inside the Viral Workshop
Put yourself in the shoes of an architect tasked with constructing a highly specialized piece of equipment; one that your survival depends upon. There’s just one catch: you have to build it inside a warehouse that wasn’t designed for your tools. The screws don’t quite fit the bolts. The instruction manuals use unfamiliar conventions. And you are very, very small, roughly 600 nanometers across.
You could try to replace everything in the building — exchange the tools, rewrite the manuals — but that would take time you don’t have. A smarter move would be to carve out a corner of the warehouse, bring the right blueprints and skilled workers together, and build a dedicated workshop.
As it turns out, scientists have discovered a giant virus that does exactly that inside the cells it infects.
Viruses are often described as freeloaders: they enter host cells as foreign entities, steal the cell’s machinery, and use it to manufacture copies of themselves. Unexpectedly, new research published in Nature Microbiology reports a more deliberate story. The authors focus on Acanthamoeba polyphaga mimivirus, a giant virus that actively remodels the interior of its host cell to build a specialized workspace for making viral proteins.
Mimiviruses are among the largest viruses ever discovered, with genomes so massive they rival those of some bacteria. They infect free-living amoebae found in soil and water. Like all viruses, mimivirus relies on its host to convert genetic instructions into proteins in a process called translation (read more here: Mimivirus – a giant in a small world).
Translation relies on ribosomes, molecular machines that “read” messenger RNA (mRNA) and assemble proteins one amino acid at a time. Each amino acid is delivered by a transfer RNA (tRNA), which matches three-letter genetic codons on the mRNA. Protein production depends on having the right translators available at the right time and in the right place.
Here’s the problem: mimivirus and its amoeba host don’t use the same dialect of the genetic code. They’re working from the same rulebook, but they favor different blueprints.
The viral genome is rich in codons ending in the nucleobases adenine (A) and uracil (U), while the amoeba’s own genes favor guanine (G) and cytosine (C). In theory, this mismatch should slow viral protein production, as the host cell has fewer tRNAs suited to viral mRNA. Yet, in practice, mimivirus thrives. Rather than flooding the cell with its own translation machinery, the virus takes a more spatial approach.
Using microscopy and ribosome-profiling techniques, the researchers found that viral mRNAs, ribosomes, and newly-synthesized proteins cluster in a distinct region surrounding the viral factory — a structure already known to be the site of viral genome replication. This region functions as a localized translation zone, in which viral mRNAs are efficiently translated into viral proteins.
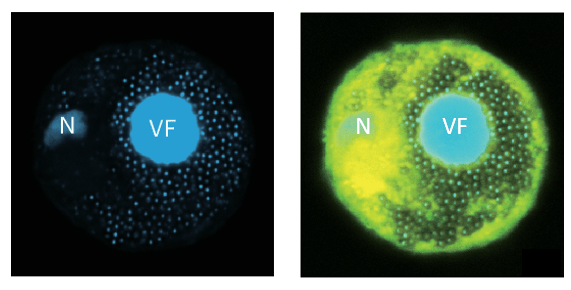
Inside this micro-environment, viral mRNAs gain preferential access to the host’s existing ribosomes and tRNAs. Ribosomes move smoothly along viral genes, showing little evidence of the stalling that typically accompanies codon mismatches. The cell’s overall tRNA population remains largely unchanged. Rather than retooling the entire warehouse, the virus simply rearranges part of it.
This finding challenges the traditional view of viral infection as diffuse and chaotic. Instead, mimivirus organizes translation in space, which is a strategy more commonly associated with complex cellular life. Mimivirus doesn’t change the tools; it changes where the work gets done.
Giant viruses already stretch the boundaries of what we consider a virus, and this study adds another layer of intrigue. Viruses can manipulate not just molecules, but cellular architecture itself. The discovery also raises new questions: how does the virus assemble this localized translation zone? Which viral and/or host proteins guide ribosomes and mRNAs to the right location? And do other viruses, or even intracellular bacteria, use similar spatial strategies?
Beyond its novelty, this research reshapes how scientists think about gene expression inside cells. Translation isn’t just about which molecules are present, but also where they are able to meet. For microbiologists, the study highlights spatial organization as an underappreciated tool in host–pathogen interactions.
Sometimes, survival isn’t about rebuilding the entire warehouse. It’s about knowing exactly where and how to set up shop.
Link to the original post: Zhang, R;, Mayer, L.; Hikida, H; et al. A giant virus forms a specialized subcellular environment within its amoeba host for efficient translation. Nat. Microbiol. (2026). https://doi.org/10.1038/s41564-025-02234-x
Additional Sources:
To read a comprehensive overview of giant viruses, click here and here.
To read another study on the mimivirus’ viral factory, click here.
Featured image: ChatGPT















