
Breaking down the microbiology world one bite at a time
Take my breath away!
Our intestines host a vast ecosystem of trillions of microscopic organisms known as gut bacteria, or the microbiome. These tiny aides digest food, create vitamins, and strengthen our defences against invaders. A groundbreaking study published in the Journal of Allergy and Clinical Immunology shows that these gut residents can predict serious lung conditions like asthma or chronic obstructive pulmonary disease (COPD) years in advance, often more accurately than traditional risk factors like smoking or age. Scientists examined stool samples from over 7,000 healthy Finnish adults collected in 2002 and tracked their medical records for up to 15 years. The results revealed strong links between specific bacterial patterns and future respiratory problems, opening new doors to early detection.
This discovery builds on a deeper connection in our bodies called the gut-lung axis, where distant organs communicate through signals carried by immune cells, chemicals, and microbial byproducts in the bloodstream. In healthy people, diverse gut bacteria support balanced inflammation, keeping airways open and functioning well. But when imbalances, known as dysbiosis occur, they can push the body toward illness. For example, low levels of helpful bacteria like Bifidobacterium or Lactobacillus raise allergy risks, while overgrowths of harmful ones like certain Clostridium species fuel ongoing inflammation. Building on this, the study used advanced DNA sequencing on fecal samples to catalog thousands of bacterial species. Machine learning algorithms then sifted through the data, spotting microbial “fingerprints” that predicted asthma onset or COPD worsening. These outperformed standard predictors: bacterial patterns nailed new asthma cases at 61% accuracy (better than demographics alone) and COPD at 78% (topping smoking history). Adding lifestyle details like body mass index (BMI), a key screening tool for body fat and health risks such as obesity, diabetes, or malnutrition, or exercise levels sharpened predictions even more, hinting at powerful early-warning tools.
To understand how this worked, consider the Finnish cohort from the larger FINRISK study. It involved 7,115 adults aged 25-74 in 2002, all starting without lung disease. Stool underwent 16S rRNA sequencing, which identifies bacteria via genetic markers, no lab culturing needed. By 2017, national health records showed 281 asthma and 258 COPD cases. Tools like random forests (groups of decision trees that vote together for better guesses) or logistic regression measured risks, revealing four bacterial groups tied to asthma and depleted anti-inflammatory microbes signaling COPD. Why focus on the gut? Early-life microbiome shapes lung defenses. C-section or formula-fed babies miss key bacteria from birth or breast milk, hiking asthma odds, much like adult patterns in the study. Fiber-fermenting bacteria produce short-chain fatty acids (SCFAs) that travel to the lungs, calming overactive immunity. The at-risk guts here matched this: fewer SCFA-makers.
These insights directly apply to asthma, a condition affecting 300 million people worldwide with airway inflammation, mucus plugs, and spasms from allergens, exercise, or stress. Wheezing, chest tightness, and coughs flare unpredictably, driven more by environment than genes. The study tied low-diversity guts to adult-onset cases, not just kid versions. Folks with “unhealthy” profiles faced 1.5-2x higher risk, regardless of allergies or weight. Imagine a 40-year-old office worker inhaling dust: their gut-primed lungs overreact. Strains like Faecalibacterium prausnitzii make butyrate, an SCFA taming asthma-triggering immune cells T-cells; drops here predicted trouble well, sparking probiotic ideas.
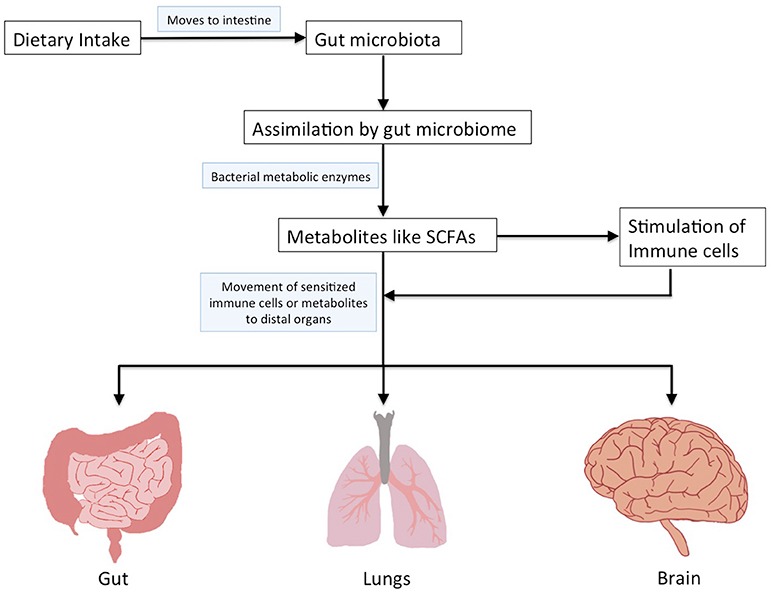
The scenario becomes much more fascinating when we go to COPD, which includes chronic bronchitis and emphysema (damaged lung air sacs that trap air and restrict breathing). Millions of people are admitted to hospitals each year as a result of long-term smoking or fumes, which permanently damage lungs and aggravate breathlessness. However, the findings disproved theories based solely on smoking: gut profiles outperformed pack-years in forecasts. Low levels of Prevotella, which breakdown fiber, have been connected to lung deterioration. Bacterial fragments such as lipopolysaccharides can enter the bloodstream through leaky intestines, causing “metabolic endotoxemia” (toxin-triggered systemic inflammation), which attacks the lungs. Viewing microorganisms holistically, that is, considering their diversity, quantity, and roles beyond individual types led to the 78% accuracy.
Beyond predictions, the study pushes for microbiome care. Plant-rich diets boost diversity, slashing asthma risk by 40% in Mediterranean styles. Fermented foods like yogurt, kimchi, or kefir deliver live bacteria, while exercise and sleep fuel them. Antibiotics wipe protectors and hence must be used sparingly. Therapeutics indicate that fecal transplants eased lung inflammation in trials by resetting guts. Tailored probiotics or prebiotics could customize prevention, with stool tests guiding high-risk groups like smokers or polluted-area dwellers.
Of course, hurdles remain. Genetics and geography vary implying not every gut predicts the same; Finns’ dairy-heavy diets differ globally, so diverse cohorts need testing. Sequencing costs limit routine checks, though cheaper shotgun metagenomics helps. The gut-lung axis touches allergies, COVID-19, even cancer dysbiosis worsened severe cases, but microbe boosts softened them. This paper sparks “microbiome medicine,” where stool foresees and forestalls disease.
Due to its huge sample size of over 7,000 and 15-year follow-up, which eliminated bias and captured actual onset, the study had good statistical power. Results remained stable after controlling for variables such as age, smoking, and socioeconomic status. Food’s significance is uncertain because no self-reported diets were found, and correlations do not establish causality; gastrointestinal patterns are linked to lung problems without proving the cause. However, cross-sectional mixtures of the past and present are inferior to prospective tracking of healthy individuals. Machine learning fueled breakthroughs, crunching 1,000+ bacterial datasets beyond human reach. Algorithms mined chaos for patterns, making elite predictions routine.
“You are what you eat” is ultimately rephrased as “your lungs reflect your gut’s diet.” Daily decisions that support healthy bacteria protect breath, which is the fundamental rhythm of life. Stool testing could soon compete with blood pressure checks, transforming covert pals into health watchdogs.
Link to the original post: Liu Y, Teo SM, Méric G, Tang HHF, Zhu Q, Sanders JG, Vázquez-Baeza Y, Verspoor K, Vartiainen VA, Jousilahti P, Lahti L, Niiranen T, Havulinna AS, Knight R, Salomaa V, Inouye M. The gut microbiome is a significant risk factor for future chronic lung disease. J Allergy Clin Immunol. 2023 Apr;151(4):943-952. doi: 10.1016/j.jaci.2022.12.810. Epub 2022 Dec 29. PMID: 36587850; PMCID: PMC10109092.
Featured image:















